Calicheamicin gamma-1 the beastly natural product – Calicheamicin gamma-1, a naturally occurring antibiotic, has emerged as a formidable weapon in the fight against cancer. Its unique structural features and potent DNA-cleaving mechanism have captivated the attention of researchers worldwide, making it a subject of intense scientific inquiry.
This comprehensive overview delves into the multifaceted nature of calicheamicin gamma-1, exploring its chemistry, biological activity, biosynthesis, and medicinal applications. We unravel the secrets of this “beastly” natural product, shedding light on its potential to revolutionize cancer treatment.
Calicheamicin Gamma-1 Overview
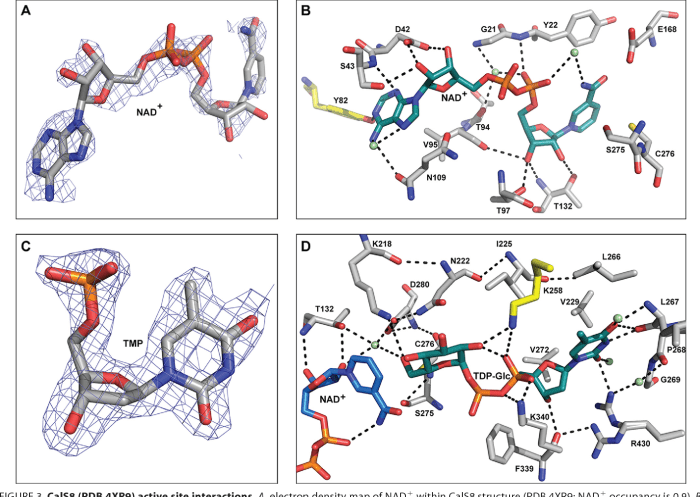
Calicheamicin gamma-1, a naturally occurring antibiotic, is a unique enediyne antibiotic renowned for its potent DNA-cleaving activity and promising applications in cancer chemotherapy.
Structurally, it comprises a macrocyclic lactam core fused to a highly reactive enediyne chromophore, endowing it with exceptional chemical reactivity and biological activity. Its molecular weight is approximately 1455 Da.
Mechanism of Action
Calicheamicin gamma-1 exerts its cytotoxic effects through a distinct DNA-cleaving mechanism.
- It binds to the minor groove of DNA, forming a DNA-calicheamicin adduct.
- The enediyne chromophore undergoes a cyclization reaction, generating a highly reactive diradical species.
- The diradical species abstracts hydrogen atoms from the DNA backbone, leading to double-strand DNA cleavage.
- The gamma-lactone ring plays a crucial role in facilitating the cyclization and DNA cleavage process.
Biological Activity and Applications
Calicheamicin gamma-1 exhibits potent cytotoxic and antitumor activities, making it a promising candidate for cancer chemotherapy.
- It has shown efficacy against a broad spectrum of cancer cell lines, including leukemia, lymphoma, and solid tumors.
- Its unique DNA-cleaving mechanism offers potential advantages over traditional chemotherapeutic agents.
- Clinical trials have demonstrated its promising antitumor activity, particularly in combination with other chemotherapeutic drugs.
Biosynthesis and Structural Analogues
Calicheamicin gamma-1 is produced by the bacterium Micromonospora echinosporavia a complex biosynthetic pathway.
- Key enzymes involved in its biosynthesis include calicheamicin synthase and enediyne synthetase.
- Structural analogues of calicheamicin gamma-1, such as calicheamicin beta-1 and calicheamicin delta-1, have been identified and characterized.
Chemical Synthesis and Modifications, Calicheamicin gamma-1 the beastly natural product
The chemical synthesis of calicheamicin gamma-1 is challenging due to its complex structure.
- Strategies for its synthesis involve biomimetic approaches and total synthesis.
- Chemical modifications have been employed to improve its stability and potency, such as glycosylation and alkylation.
Medicinal Chemistry and Drug Development
Calicheamicin gamma-1 has inspired the design of new anticancer agents with improved properties.
- Structure-activity relationship studies have identified key structural features responsible for its biological activity.
- Ongoing research and development efforts aim to optimize its therapeutic potential and address limitations.
Essential FAQs: Calicheamicin Gamma-1 The Beastly Natural Product
What is the molecular weight of calicheamicin gamma-1?
Approximately 1175 g/mol
How does calicheamicin gamma-1 cleave DNA?
It forms a covalent adduct with DNA, leading to strand breakage through a unique mechanism involving a gamma-lactone ring.
What are the potential applications of calicheamicin gamma-1 in cancer therapy?
It has shown promise in treating various cancers, including leukemia, lymphoma, and solid tumors.